The Bulk lyophilization services play a vital role in the preservation and stabilization of sensitive pharmaceutical products, particularly biologics and vaccines. These services enable the transformation of liquid drug substances into stable, dry formulations that can be stored and transported efficiently. With the growing complexity of drug development pipelines, freeze drying technologies have become essential for ensuring product integrity, longer shelf life, and reduced dependency on stringent cold chain systems.
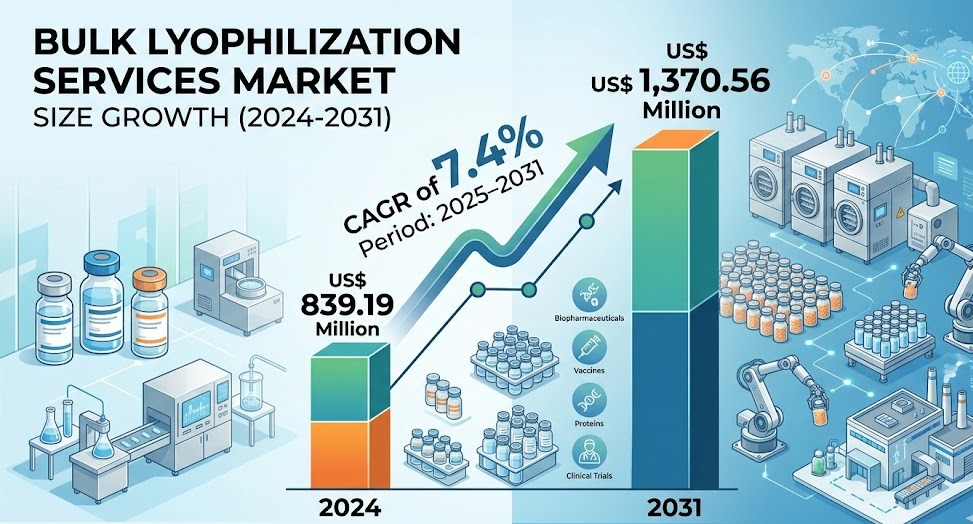
The Bulk Lyophilization Services Market Scope continues to expand significantly, supported by rising biologics demand and increased outsourcing to specialized service providers. The industry is projected to grow from US$ 839.19 million in 2024 to US$ 1,370.56 million by 2031, registering a CAGR of 7.4% during 2025 to 2031. This growth reflects the widening scope of services, technological innovations, and global adoption across pharmaceutical manufacturing processes.
Expanding Scope Across Technologies and Formats
The scope of bulk lyophilization services covers a wide range of technologies and formats tailored to different production requirements. Bulk tray lyophilization holds a leading share due to its scalability and efficiency in handling large volumes of biologics. Other formats such as drum lyophilization, shelf freeze drying, and tunnel based systems provide flexibility for continuous and specialized drying applications.
Technological advancements are further expanding the service scope. Innovations such as controlled ice nucleation, quality by design frameworks, and process analytical technology enhance process precision and product consistency. These technologies help reduce cycle times while maintaining uniform drying, which is critical for large scale pharmaceutical production. Hybrid methods that combine traditional and advanced approaches are also gaining traction, especially for complex biologics and gene based therapies.
Download Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00041488
Broad Service Scope Across Development and Commercialization
The bulk lyophilization services industry encompasses a comprehensive range of services from early stage development to full scale commercial manufacturing. Custom process development and optimization services are essential for designing stable formulations and ensuring regulatory compliance. Pilot scale lyophilization supports clinical research and small batch production, enabling pharmaceutical companies to test and refine their products.
Full scale commercial bulk lyophilization accounts for the largest share within the service scope, driven by increasing demand for high volume production of biologics and vaccines. In addition, the scope extends across multiple operational scales including lab, pilot, and industrial levels. Commercial and industrial scale services are witnessing rapid growth as pharmaceutical companies expand production capacity to meet global demand.
End User Scope and Emerging Applications
Pharmaceutical and biotechnology companies represent the primary end users within the scope of bulk lyophilization services. These organizations rely heavily on freeze drying technologies to stabilize products such as monoclonal antibodies, vaccines, and advanced therapeutics. Research and academic institutions also contribute to demand, particularly in early stage innovation and drug development.
The scope is further expanding into emerging applications such as cell and gene therapies. These advanced treatments require highly specialized preservation techniques to maintain their effectiveness. As the number of clinical trials in these areas continues to increase, demand for customized and small volume bulk lyophilization solutions is expected to rise. Additionally, the growing preference for lyophilized injectables is enhancing the relevance of these services across modern healthcare systems.
Regional Scope and Global Market Expansion
The geographic scope of bulk lyophilization services spans North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. North America leads the global landscape, supported by strong biopharmaceutical research activities, advanced manufacturing infrastructure, and the presence of numerous contract development and manufacturing organizations.
Asia Pacific is emerging as the fastest growing region, driven by expanding pharmaceutical manufacturing capacity, government support, and cost advantages. Countries such as India and China are becoming key hubs for outsourced production, contributing to the global expansion of lyophilization services. Europe also maintains a significant share due to its strong regulatory framework and early adoption of advanced freeze drying technologies.
Emerging regions including Latin America and the Middle East and Africa are gradually strengthening their position through investments in healthcare infrastructure and local vaccine production. This regional diversification is broadening the overall scope and accessibility of bulk lyophilization services worldwide.
Competitive Landscape and Key Players
The competitive landscape is characterized by the presence of both global leaders and specialized regional players. Companies are focusing on expanding production capacity, adopting advanced technologies, and forming strategic partnerships to enhance their service offerings.
Key players operating in the industry include:
- ATTWILL Medical Solutions
- PCI Pharma Services
- Lyophilization Technology, Inc.
- OFD Life Sciences
- Symbiosis Pharmaceutical Services Ltd
- Quality BioResources
- Affinity Life Sciences, Inc.
- Alcami Corporation
- Hudson Valley Lyomac
- Catachem Inc.
These companies are actively investing in automation, real time monitoring systems, and process optimization to improve efficiency and meet growing demand.
Future Outlook
The future outlook for bulk lyophilization services remains highly promising as the pharmaceutical industry continues to evolve. Increasing production of biologics, rising demand for vaccines, and growing adoption of advanced therapies will sustain market growth. Technological innovations such as artificial intelligence driven monitoring, continuous manufacturing integration, and energy efficient processes are expected to further enhance operational efficiency.
Related Reports @
Bupivacaine Liposome Injectable Suspension Market Drivers, Trends, and Forecasts by 2031
Sutherlandia Extract Market Research Report by Size Share Analysis 2031
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean| German| Japanese| French| Chinese| Italian| Spanish